Recombinant Troponin T
Recombinant Troponin T - Lyophilized | Recombinant Troponin T - Liquid
(View Recombinant Troponin T Data as PDF)
Recombinant Human Troponin T (TnT) from Scripps Laboratories is available for research and diagnostic assay development. Native TnT was used by the diagnostic industry for decades, but it is unstable and its purification requires a lengthy manufacturing protocol. In addition, the amount of TnT present in the starting material can vary widely. These factors make native TnT an expensive protein to purify and a challenging one to stabilize.
Scripps Laboratories offers highly purified Recombinant TnT as a reliable and economical alternative to native TnT. The data presented here demonstrate its suitability for research use, immunoassay development, and large-scale diagnostic assay manufacturing.
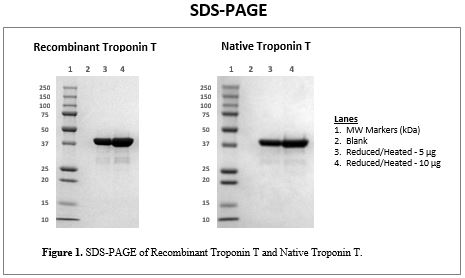
SDS-PAGE
Figure 1 presents SDS-PAGE images of highly purified Recombinant TnT and Native TnT. Both proteins display an approximate molecular weight of 36 kDa, which is consistent with the reported value.1 In addition, minor bands are visible in the reduced/heated and non-reduced/not heated samples of the recombinant and native products. In the western blot images that follow, these bands are confirmed to be molecular weight variants of TnT.
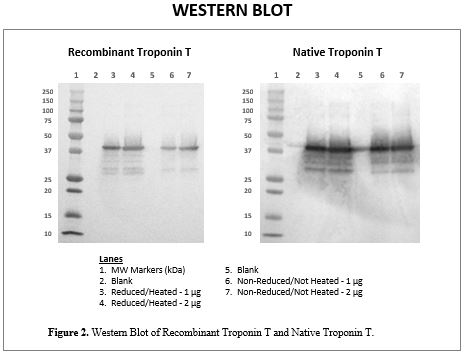
WESTERN BLOT
TnT is known to be unstable in human plasma or serum and as a purified reagent. For this reason, it is often provided in lyophilized form or, if it is supplied as a liquid, it is presented in a buffer with a high concentration of urea. This instability leads to multiple molecular weight bands, which are visible in the SDS-PAGE shown in Figure 1. Upon Western Blot, the identity of these bands is confirmed to be Troponin T, using a highly specific, anti-TnT monoclonal antibody. This is demonstrated in Figure 2, with the blots of both Recombinant TnT and Native TnT.
The data presented here for Recombinant TnT show it to be an excellent alternative to native TnT. Produced without the use of affinity tags in E. coli, Recombinant TnT is available in large, bulk lots with excellent lot-to-lot consistency.
Recombinant Human Troponin T is in stock and available now in lyophilized and liquid form:
Recombinant Troponin T, lyophilized
Recombinant Troponin T, liquid
References
(1) Marston and Zamora. J. Muscle Res. Cell Motil., 2020, (41): 71-89.
